Lösung von Aufgabe 16
-
thermische Größen:
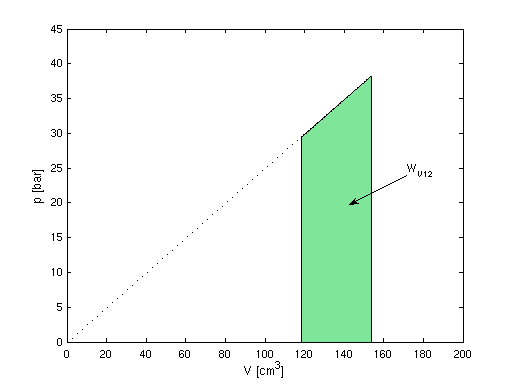
- p1 = 29.5 bar, T1 = 1218
K
- V1 = m Ri T / p = m R T / (M
p) = 118.6 cm3
- p2 = 38.2 bar
- pV -1 = const ⇒ V2 =
V1 p2 / p1 = 153.6
cm3
- TV -2 = const ⇒ T2 =
T1 V22 / V12
= 2043 K
-
p-V-Diagramm:
-
kalorische Größen:
- WV12 = (m Ri)/(n - 1)
(T2 - T1) = - 118.4 J
- H2 - H1 = m
 p (T2 -
T1) = 1006 J
p (T2 -
T1) = 1006 J
- U2 - U1 = m
 V (T2 -
T1)
V (T2 -
T1)
- = m (
 p - Ri)
(T2 - T1) = 769.0 J
p - Ri)
(T2 - T1) = 769.0 J
- Verbrennungswärme:
- Q12 = U2 - U1 -
WV12 = 887.4 J



 p (T2 -
T1) = 1006 J
p (T2 -
T1) = 1006 J V (T2 -
T1)
V (T2 -
T1) p - Ri)
(T2 - T1) = 769.0 J
p - Ri)
(T2 - T1) = 769.0 J