Lösung von Aufgabe 17
-
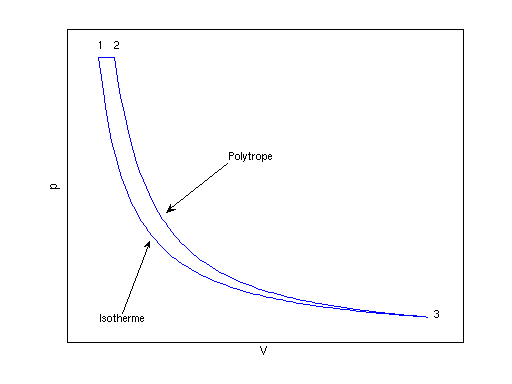
p-V-Diagramm:
-
Zustandsgrößen:
-
Punkt 1
- p1 = 150 bar, T1 = 313.15
K
- V1 = m Ri T / p = 0.5996
m3
- U1 = 0
- H1 = U1 + p1
V1 = 8.994 MJ
-
Punkt 2
- p2 = 150 bar
- Q12 = m cp
(T2 - T1)
- ⇒ T2
= Q12/(m cp) + T1 = 811.2 K
- V2 = V1 T2 /
T1 = 1.553 m3
- U2 = m cv
(T2 - T1) + U1
- = m (cp -
Ri) (T2 - T1) + U1
= 35.70 MJ
- H2 = U2 + p2
V2 = 58.99 MJ
-
Punkt 3
- T3 = T1 = 313.15 K
- Aus pn-1 / Tn = const.
folgt:
- p3 = p2
(T3/T2) n/(n - 1) = 0.4966
bar
- V3 = m Ri T3
/ p3 = 181.1 m3
- U3 = U1 = 0
(Isotherme!)
- H3 = U3 + p3
V3 = 8.994 MJ
-
Nutzarbeit Wk = Σ WVi
- WV12 = p1 (V1 -
V2) = - 14.30 MJ
- WV23 = (m Ri)/(n - 1)
(T3 - T2) = - 71.51 MJ
- WV31 = p3 V3
ln(p1/p3) = 51.36 MJ
- Wk = - 34.46 MJ
-
thermischer Wirkungsgrad:
- η = (|Wk|)/(Q12 +
Q23)
- Q12 = 50 MJ
- Q23 = WV23 (n -
κ)/(κ - 1) = 35.82 MJ
- ⇒ η = 0.4015
- ηCarnot = 1 -
T3/T2 = 0.6139