Lösung von Aufgabe 20
-
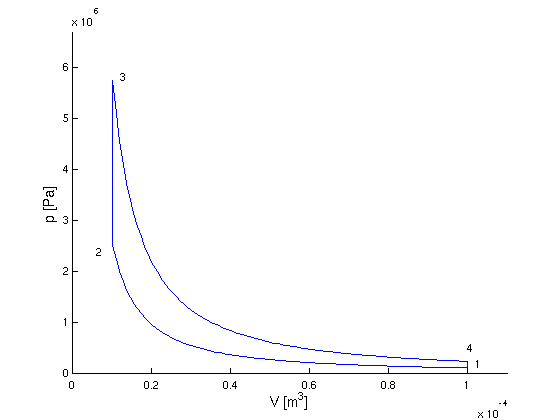
p-V-Diagramm:
-
Thermische Zustandsgrößen:
-
Punkt 1
- p1 = 1 bar, V1 = 100
cm3, T1 = 343 K
-
Punkt 2 (längs Isentrope):
- V2 = 10 cm3
- p2 = p1
(V1/V2)κ = 25.12
bar
- T2 = T1
(V1/V2)κ - 1 = 861.6
K
-
Punkt 3 (längs Isochore):
- T3 = 1973 K
- V3 = V2 = 10
cm3
- p3 = p2
(T3/T2) = 57.52 bar
-
Punkt 4 (längs Isentrope):
- V4 = V1 = 100
cm3
- p4 = p3
(V3/V4)κ = 2.290 bar
- T4 =
T3(V3/V4)κ -
1 = 785.5 K
-
Wirkungsgrad:
- η = -Wk / Q23 =
(Q23 + Q41)/Q23
- = 1 +
Q41/Q23
-
Wegen angenommener Temperaturunabhängigkeit von
cV:
- Q23 = m cV
(T3 - T2)
- Q41 = m cV
(T1 - T4)
- ⇒ η = 1 +
(T1 - T4)/(T3 - T2)
= 0.6019
- ηCarnot = 1 -
T1/T3 = 0.8262